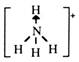Acids bases and salts
Acids bases and salts
The following texts are the property of their respective authors and we thank them for giving us the opportunity to share for free to students, teachers and users of the Web their texts will used only for illustrative educational and scientific purposes only.
The information of medicine and health contained in the site are of a general nature and purpose which is purely informative and for this reason may not replace in any case, the council of a doctor or a qualified entity legally to the profession.
![]()
Acids bases and salts
Chemistry
ACIDS, BASES, and SALTS
Chapter Outline
- Arrhenius Definitions of Acids and Basses
- Brønsted-Lowry’s Definitions of Acids and Bases
- Brønsted-Lowry’s Concept of Acid-Conjugate Base Pairs
- Naming Binary and Oxo-Acids
- Is Water an Acid or a Base?
- The pH Scale
- Calculations of pH of solutions of strong acids and strong bases.
- Relative Strength of Acids and Bases
- Analyzing Acids and Bases
- Acid-Base Titration and Calculations Involving Titration Data
- Hydrolysis Reactions of Salts
- Buffer Solutions
Before the 20th Century, acids, bases and salts are characterized by properties such as taste and their ability to change the color of litmus (an organic dye). Acids taste sour, bases taste bitter, and salts are salty. Acids turn blue litmus to red, while bases turn red litmus to blue.
Arrhenius Definitions:
- Acids are substances that dissociate in water to produce hydrogen ions (H+).
- Bases are substances that dissociate to produce hydroxide ions (OH-).
Examples:
HCl is an acid: HCl(aq) à H+(aq) + Cl-(aq);
NaOH is a base: NaOH(aq) à Na+(aq) + OH-(aq); è NaOH is a base.
Brønsted-Lowry Definitions:
- Acids are substances that donate/lose a proton in chemical reactions;
- Bases are substances that accept/gain a proton in chemical reactions.
Examples of Bronsted-Lowry acid-base reactions:
H:A + B: ß à B:H+ + A-
Acid1 base2 conjugate conjugate
Acid2 base1
(conjugate acid-base pairs are HA-A- and B:H+-B:)
HCl(aq) + H2O ß à H3O+ + Cl-
Acid1 base2 conjugate conjugate
Acid2 base1
(Conjugate acid-base pairs: HCl-Cl- and H3O+-H2O)
NH4+(aq) + OH-(aq) ß à H2O + NH3(aq)
Acid1 base2 conjugate conjugate
Acid2 base1
(conjugate acid-base pairs: H2O-OH- and NH4+-NH3)
Exercise-1: Identify all Brønsted acids, bases, and their acid-conjugate base pairs:
a. HC2H3O2(aq) + H2O ßà H3O+(aq) + C2H3O2-(aq);
b. CH3NH2(aq) + H2O ßà CH3NH3+(aq) + OH-(aq);
c. H2SO4(aq) + H2O ßà H3O+(aq) + HSO4-(aq);
d. H2PO4-(aq) + H2O ßà H3O+(aq) + HPO42-(aq);
_________________________________________________________________________
Naming Acids
There are TWO types of acids:
- Binary acids (acids that do not contain oxygen atom);
- Oxo-acids (those containing oxygen atoms in their formula)
The names of binary acids start with hydro-, followed by the first syllable of the anion's name, and end with -ic:
HF - hydrofluoric acid; HCl - hydrochloric acid; HBr - hydrobromic acid;
HI - hydroiodic acid; H2S - hydrosulfuric acid; HCN - hydrocyanic acid;
The names of oxyacids are derived from the name of oxy-anion the acids contain. For an anion whose name ends with -ate, the acid’s name starts with the first syllable of the anion and ends with –ic. If the anion’s name ends with -ite, the name of acid starts with the first syllable of the anion's name and ends with -ous.
Anions Anion’s Names Acids Names of Acids___
NO3- nitrate ion HNO3 nitric acid
NO2- nitrite ion HNO2 nitrous acid
SO42- sulfate ion H2SO4 sulfuric acid
SO32- sulfite ion H2SO3 sulfurous acid
PO43- phosphate ion H3PO4 phosphoric acid
C2H3O2- acetate ion HC2H3O2 acetic acid
ClO- hypochlorite HClO hypochlorous acid
ClO2- chlorite HClO2 chlorous acid
ClO3- chlorate HClO3 chloric acid
ClO4- perchlorate HClO4 perchloric acid
____________________________________________________
Note that, the ionizable hydrogen in oxo-acids is bonded to the oxygen in the molecule.
Strong and Weak Acids
Strong acid – one that ionizes completely in aqueous solution;
Examples of Strong Acids: HClO4, HCl, H2SO4, HNO3, HBr, and HI
Weak acid – one that do not ionize completely in aqueous solution;
Examples of weak acids: HC2H3O2, HF, HNO2, HClO, HClO2, H2SO3, H2CO3, and H3PO4.
Strong bases also ionize completely when dissolved in water.
Examples: NaOH, KOH, and Ba(OH)2
Weak bases do not ionize completely when dissolved in water.
Examples: NH3 (or NH4OH), NH2OH, Mg(OH)2, and all hydroxides and oxides that are only slightly soluble in water.
What determines acid strength?
Consider the following reversible process when an acid is dissolved in water:
HA(aq) + H2O ßà H3O+(aq) + A-(aq)
In the forward reaction, acid HA donates a proton to water molecule to form hydronium ion, H3O+, and the conjugate base A-. Water acts as a Bronsted-Lowry’s base.
- The acid strength is measured in terms of their degree of ionization (or dissociation) in water. If an acid ionizes completely, it is considered a strong acid.
- A strong acid has a weak conjugate base - the conjugate base loses its proton to water quite readily. It is less willing to compete for a proton with water. The stronger the acid, the weaker is its conjugate base.
- The equilibrium for strong acid shifts far to the right. The acid ionization constants, Ka, for strong acids are very large.
A weak acid does not readily gives up its proton to water. Its conjugate base is a stronger base than water. The weaker the acid, the stronger is its conjugate. The ionization equilibrium for weak acids shifts far to the left.
- Strong acids have weak conjugate bases and weak acids have strong conjugate bases.
Cl-, Br-, I-, ClO4-, HSO4-, and NO3- are weak conjugate bases;
C2H3O2-, F-, CN-, NO2-, HSO3-, SO32-, H2PO4-, HPO42-, PO43- are strong conjugate bases;
An aqueous solution of strong acid contains only hydronium ions, H3O+, and its conjugate bases. For example, in HCl(aq) solution there are only H3O+ and Cl- ions, but no HCl molecules. While an aqueous solution of weak acid, such as acetic acid, contains mainly the undissociated molecules, HC2H3O2, with a small fraction (~1%) of H3O+ and C2H3O- ions.
Exercise-2:
1. Select all acids that have strong conjugate bases?
(a) HNO3 (b) H3PO4 (c) HClO (d) HBr (e) H2S
2. Select all acids that have weak conjugate bases?
(a) HF (b) HClO4 (c) HI (d) HCN (e) H2SO4
________________________________________________________________________
Is Water as an Acid and a Base?
Water may acts as an acid or a base. It is an amphoteric substance – one that can act as an acid or a base.
Ionization of water: 2H2O ßà H3O+(aq) + OH-(aq);
The equilibrium constant expression is, K = [H3O+][OH-]
[H2O]2
k[H2O]2 = Kw = [H3O+][OH-] = 1.0 x 10-14 at 25oC
Kw is called ion-product constant for water.
In pure water, the molar concentration, [H3O+] = [OH-] = √(1.0 x 10-14) = 1.0 x 10-7 M.
(For simplicity we will use the concentration symbol [H+] for [H3O+])
If [H+] = [OH-] = 1.0 x 10-7 M => the solution is said to be neutral;
If [H+] > 1.0 x 10-7 M, [OH-] < 1.0 x 10-7 M, => solution is acidic ([H+] > [OH-])
If [H+] < 1.0 x 10-7 M, [OH-] > 1.0 x 10-7 M, => solution is basic ([H+] < [OH-])
The pH Scale
The pH scale is used to measure the acidity (or basicity) of a solution, especially when the hydrogen ion concentration is very low.
pH = - log[H+]. Likewise, pOH = - log[OH-]
In neutral solutions, [H+] = 1.0 x 10-7 M, è pH = - log(1.0 x 10-7) = 7.00;
Neutral solutions also contains [OH-] = 1.0 x 10-7 M, è pOH = - log(1.0 x 10-7) = 7.00
In acidic solutions, [H+] > 1.0 x 10-7 M, and pH < 7.00;
while in basic solutions, [H+] < 1.0 x 10-7 M, and pH > 7.00.
Thus, pH = 7 è a neutral solution;
pH < 7 è an acidic solution, and
pH > 7 è a basic solution
(Note: Kw = [H+][OH-] = 1.0 x 10-14; è pKw = - log(Kw) = - log(1.0 x 10-14) = 14.00
But, pKw = pH + pOH = 14.00; è pOH = 14.00 - pH, and pH = 14.00 - pOH;
When pH = 7 è pOH = 7; pH < 7 è pOH > 7, and pH > 7 è pOH < 7;
Thus, pH < 7 è [H+] > [OH-]; and pH > 7 è [OH-] > [H+]
Examples: When [H+] = 1.0 x 10-4 M, pH = - log(1.0 x 10-4) = 4.00;
When [OH-] = 1.0 x 10-4 M, [H+] = 1.0 x 10-14 = 1.0 x 10-10 M; pH = 10.00
1.0 x 10-4 M
Alternatively, [OH-] = 1.0 x 10-4 M è pOH = -log[OH-] = -log(1.0 x 10-4 M) = 4.00;
è pH = 14.00 – 4.00 = 10.00
Quick Reference for [H+] and pH
—————————————————————————————————————
[H+], M pH [H+], M pH
———————————— ——————————————
1.0 x 10-1 1.00 1.0 x 10-8 8.00
1.0 x 10-2 2.00 1.0 x 10-9 9.00
1.0 x 10-3 3.00 1.0 x 10-10 10.00
1.0 x 10-4 4.00 1.0 x 10-11 11.00
1.0 x 10-5 5.00 1.0 x 10-12 12.00
1.0 x 10-6 6.00 1.0 x 10-13 13.00
1.0 x 10-7 7.00 1.0 x 10-14 14.00
——————————————————————————————————————
Acis and The Hydrogen Ion Concentration:
For strong monoprotic acids (those containing one ionizable hydrogen atom), such as HCl, HNO3, and HClO4, [H+] = [HX]0 (initial acid concentration)
For example, a solution of 0.10 M HCl has [H+] = 0.10 M, and the pH of solution is 1.00
Since weak acids dissociate only partially, the [H+] << [HX]0 (initial acid concentration).
For example, a solution of 0.10 M HF has [H+] << 0.10 M and pH > 1.00
Strong bases also dissociate completely and produce a high concentration of OH-. For strong bases such as NaOH and KOH, which contain one equivalence of OH- per mole, the OH- concentration is the same as the initial base concentration. For example, a 0.10 M NaOH solution contains [OH-] = 0.10 M. The pH of this solution is 14.00.
A strong base such as Ba(OH)2, which contains two equivalence of OH- per mole, the OH- concentration is equal to twice the initial base concentration. That is, a 0.10 M Ba(OH)2 solution has [OH-] = 0.20 M and the pH of 0.10 M Ba(OH)2 is 14.70
In weak bases the [OH-] is much less than the initial concentration of the base dissociate only partially. For example, a 0.10 M NH3 solution has [OH-] << 0.10 M. The pH of 0.10 M NH3 solution < 14.00
Exercise-3:
1. Calculate [H+] and [OH-] in the following solutions, and determine the pH and pOH, respectively, of each solution. State whether the solution is neutral, acidic or basic.
(a) 2.0 x 10-4 M HCl
(b) 2.0 x 10-5 M NaOH
(c) 2.5 x 10-4 M Ba(OH)2
2. What is the concentration [H+] in a solution with pH = 4.75.
3. What is the concentration [OH-] in a solution with pH = 10.50
_________________________________________________________________________
Preparing Dilute Acid Solutions
Most common acids, such as HCl(aq), HNO3, H2SO4, and HC2H3O2 are procured as concentrated solutions. For examples, HCl(aq) comes in 12 M, HNO3 in 16 M, H2SO4 in 18 M, and HC2H3O2 in 17.5 M. Lower acid concentrations are prepared by dilution of concentrated acid solutions. For example, to prepare 1.00 L solution of 1.5 M HCl from the concentrated (12 M) HCl, we would need to know how much of the concentrated acid must be diluted. This is done by calculating the number of moles of HCl needed in the final (dilute) solution as follows:
Mole HCl in final solution = 1.00 L solution x (1.5 mol/L solution) = 1.5 mole HCl
We can then calculate the volume of concentrated acid that contains the same mole of HCl, such as,
? L of conc. HCl x (12 mol/L solution) = 1.5 mole HCl
? L of conc. HCl = 1.5 mole HCl x (L HCl/12 mol HCl) = 0.125 L = 125 mL.
That is, 125 mL of 12 M HCl is needed to make 1.00 L of 1.5 M HCl solution. We can prepare this solution in a 1.00-L volumetric flask as follows:
- The volumetric flask is first filled with de-ionized water to about a-third full,
- 125 mL of conc. HCl is carefully measured and transferred quantitatively into the flask,
- after initial mixing, more de-ionized is added until the water meniscus comes to the 1.00-L mark on the neck of the flask.
- The solution is thoroughly mixed by inverting the stoppered flask several times.
(A simple equation that can be used in dilution method is, M1V1 = M2V2; where subscript-1 implies initial solution and subscript-2 implies final solution.)
Exercise-4:
1. Describe how you would prepare 500. mL of 0.45 M NaOH solution from 6.0 M NaOH.
2. How many milliliters of 15 M NH3 are needed to prepare 2.00 L of 3.0 M aqueous ammonia solution?
3. If 75.0 mL of 6 M HCl(aq) is diluted with de-ionized water to make 1.5 L solution, what is the concentration of the dilute acid?
——————————————————————————————————————
Volumetric Analyses by Acid-Base Titration
Volumetric analysis is a method of determining the concentration of one solution by titration using another solution which concentration and the reaction stoichiometry are known. Titration is a technique of carrying out a reaction involving solutions such that their exact volumes are known. One of the solutions (usually the acid) of known volume is placed in an Erlenmeyer flask or beaker and the base solution is added from a buret until the end-point is reached. The titration requires the use of a suitable acid-base indicator, which is added to the acid solution in the flask (or beaker). An acid-base indicator is an organic substance that exhibits a different colors in acidic and basic solution.
The end-point of titration is indicated by a change in the indicator color. If the volume and concentration of the acid are known, its number of moles can be calculated. From the reaction stoichiometry (from the balanced equation), the number of moles of the base and its concentration can be determined.
Calculations of Molar Concentration from Acid-base Titration.
When an acid such as HCl(aq) is titrated with aqueous NaOH, the following reaction occurs:
HCl(aq) + NaOH(aq) à NaCl(aq) + H2O;
The stoichiometric ratio of HCl to NaOH is 1 mole HCl to 1 mole NaOH. In titration, the volume and concentration of the standard solution (acid or base) are known, but only the volume of the other solution (acid or base) whose concentration to be determined is known. The above stoichiometry enables us to calculate the unknown concentration.
Example-1: suppose that 25.00 mL of 0.2250 M HCl(aq) is required to neutralize 27.45 mL of aqueous NaOH solution whose concentration is not known. The mole of each reactant and the concentration of NaOH can be calculated as follows:
No. of mol of HCl reacted = 25.0 mL x (1 L/1000 mL) x (0.2250 mol/L) = 0.005625 mol.
Since HCl and NaOH react in a 1:1 ratio,
No. of mol of NaOH = No. of mol of HCl = 0.005625 mol;
Molarity of NaOH = (0.005625 mol/0.02745 L) = 0.2049 M
Example-2: 20.0 mL H2SO4 of unknown concentration requires 32.0 mL of 0.205 M NaOH. Calculate the concentration of H2SO4 solution. The reaction between sulfuric acid and NaOH is
H2SO4(aq) + 2 NaOH(aq) à Na2SO4(aq) + 2 H2O
The stoichiometric ratio is 1 mole of H2SO4 to 2 moles of NaOH. In the above example,
Mol of NaOH reacted = 0.205 mol. NaOH x 32.0 mL x 1 L___ = 0.00656 mol;
1 L solution 1000 mL
Mol of H2SO4 = Mol NaOH x (1 mol H2SO4/2 mol NaOH)
= 0.00656 mol NaOH x (1 mol H2SO4/2 mol NaOH) = 0.00328 mol
Molarity of H2SO4 = 0.00328 mol H2SO4 = 0.164 M
0.0200 L
Exercise-5:
1. In an acid-base titration, 25.0 mL of 0.204 M HCl(aq) requires 22.5 mL of NaOH(aq) to reach end-point. What is the concentration of NaOH(aq)?
2. A 10.0-mL sample of a brand of vinegar is titrated with 0.225 M NaOH(aq) solution, which requires 36.0 mL of the base to reach end-point. What is the molar concentration of acetic acid in the vinegar? The reaction is
HC2H3O2(aq) + NaOH(aq) à NaC2H3O2(aq) + H2O
3. A 20.00-mL solution of barium hydroxide, Ba(OH)2(aq), is found to require 28.40 mL of 0.1980 M HCl(aq) to neutralize the base. Write a balanced equation for the reaction of barium hydroxide with hydrochloric acid and calculate the molar concentration of the base.
___________________________________________________________________________
Salts and the Properties of Salt Solutions
Salts are products of acid-based reactions. For example, NaCl is a product of the following acid base reaction:
HCl(aq) + NaOH(aq) à NaCl(aq) + H2O
In the chemical formula of a salt, the cation is contributed by the base, while the anion is contributed by the acid in the acid-base reaction.
Four different types of salts:
- Salts of strong acid-strong base reactions (e.g. NaCl, KNO3, NaClO4, etc.)
- Salts of weak acid-strong base reactions (e.g. NaC2H3O3, K2CO3, KCN, NaCHO2, etc.)
- Salts of strong acid-weak base reactions (e.g. NH4Cl, NH4NO3, HONH3Cl, etc.)
- Salts of weak acid-weak base reactions (e.g. NH4C2H3O2, NH4CN, NH4HS, etc.)
These different types of salts when dissolved in water will produce either acidic, basic, or neutral solutions. Salts derived from strong acid-strong base reaction will yield a neutral solution; for example, aqueous solutions of NaCl, KNO3, NaClO4, etc. are neutral.
Salts from weak acid-strong base reactions will form basic solutions; for example, aqueous solutions of NaC2H3O2, K2CO3, Na3PO4, etc. are basic. The basic nature of the salt solution is due to the hydrolysis reactions of the strong conjugate base of the weak acid. These strong conjugate bases can compete for the H+ ions in water. For example,
NaC2H3O2(aq) à Na+(aq) + C2H3O2-(aq); (dissociation of ionic compound)
C2H3O2-(aq) + H2O ßà HC2H3O2(aq) + OH-(aq)
The second reaction, called hydrolysis reaction, produces OH- ions and a basic solution.
Salts that are formed from strong acid-weak base reactions are acidic; for example, aqueous solutions of NH4Cl and NH4NO3 are acid. For example,
NH4Cl(aq) à NH4+(aq) + Cl-(aq)
NH4+(aq) + H2O ßà NH3(aq) + H3O+(aq)
The hydrolysis reaction of NH4+ produces H3O+, which makes the solution acidic. Finally, salts of weak acid-weak base reactions may be acid, basic, or neutral, depending on the relative strength of NH4+ as an acid and the respective anion as a base. For example, aqueous solution of ammonium acetate, NH4C2H3O2, is neutral, whereas aqueous solution of ammonium phosphate, (NH4)3PO4, is basic, and ammonium nitrite, NH4NO2 solution is slightly acidic.
Exercise-6:
1. Predict whether each of the following salt solutions is expected to be neutral, acid, or basic:
(a) NH4ClO4, (b) NaF (c) KI (d) NH4NO3 (e) Na2CO3
________________________________________________________________________
Buffer Solution
- A solution that is able to maintain its pH (with very little change) even when a little amount of strong acid or strong base is added to it.
Consider the following cases:
When 0.01 mol of HCl is added to 1 L of pure water, [H+] increases from 10-7 to 10-2 M, and the pH changes from about 7 to 2, which implies that water is not a buffer.
When the same amount of HCl is added to a solution containing a mixture of 1 M acetic acid (HC2H3O2) and 1 M sodium acetate (NaC2H3O2), the pH of the solution changes very little – the pH changes from 4.74 to about 4.66. Therefore, a solution that is composed of acetic acid and sodium acetate is a buffer solution.
Buffer solutions are made up of a mixture of weak acid and the salt that contains the conjugate base of the acid in fair amounts to provide a buffering capacity. It can also be made from a mixture of weak base and the salt that has the conjugate acid to the weak base, such as a mixture of NH3 and NH4Cl in fair amount.
Some examples of buffer solutions are:
HC2H3O2-NaC2H3O2, HCHO2-NaCHO2, H2CO3-NaHCO3, KH2PO4-K2HPO4,
H2SO3-NaHSO3, and NH4Cl-NH3(aq).
Buffered solutions are vital to living organisms. All metabolic reactions are controlled or accelerated by biological catalysts called enzymes, which function only within a narrow pH range. For example, it is important that our body fluid be maintained at a certain (narrow) pH range. Our blood is maintained at the pH range of 7.30 - 7.40. A drop to below pH 7 or a rise to above pH 7.5 can be fatal.
What types of buffering reactions occur in a buffered solution?
Consider a buffered solution composed of KH2PO4 and K2HPO4. The species present in solution are primarily K+, H2PO4- and HPO42-. (K+ is a spectator ion and not involved in the buffering reaction.)
When a little strong acid is added to this solution, the H+ ions from the acid reacts with the base component of the buffer (in this case HPO42-):
H+(aq) + HPO42-(aq) à H2PO4-(aq) ….(buffering reaction-1)
When a strong base, such as NaOH is added, the OH- reacts with the acid component of the base (in this case H2PO4-):
OH-(aq) + HC2H3O2(aq) à H2O + C2H3O2-(aq) ….(buffering reaction-2)
Reactions (1) and (2) are important buffering reactions that maintain the pH of the solution. Two buffer systems – the phosphate buffer, H2PO4--HPO42-, and carbonic acid-bicarbonate buffer, H2CO3-HCO3-, are important buffer systems that maintain the normal blood pH. The buffering reactions of bicarbonate buffer are:
H+(aq) + HCO3-(aq) à H2O + CO2(aq);
OH-(aq) + CO2(aq) + H2O à 2 HCO3-(aq)
Summary of Characteristics of buffer solutions:
1. The solution contains a weak acid HX and its conjugate base X-, or a weak base B and its conjugate acid BH+, in fair amount;
2. A buffer solution maintains its pH by absorbing excess H+ or OH- produced by a strong ac id or strong base, so that these ions do not accumulate.
3. The buffering reactions involve the reaction of H+ with the conjugate base X- in the buffer or the reaction of OH- with the acid component (HX) of the buffer:
H+(aq) + X-(aq)à HX(aq); OH-(aq) + HX(aq) à H2O + X-.
These two reactions prevent a significant increase in [H+] or [OH-] in the solution.
4. The buffering capacity of a buffer solution implies the amount of H+ or OH- it can absorb without significantly changing its pH. Buffering capacity depends on the concentration of the weak acid and its conjugate base in the solution.
5. The buffering range of a buffer solution depends on the pKa of the acid component of the buffer. A given buffer will buffer best in the pH range = pKa ± 1.
Source: http://vistawww.peralta.edu/Projects/10040/Chem%2030A/Chapter9_Acids,%20Bases,%20and%20Salts.doc
Web site: http://vistawww.peralta.edu/
Author : not indicated on the source document of the above text
Google key word : Acids bases and salts file type : doc
If you are the author of the text above and you not agree to share your knowledge for teaching, research, scholarship (for fair use as indicated in the United States copyrigh low) please send us an e-mail and we will remove your text quickly.
Acids, Bases and Salts 10
It is often said that love makes the world go around. Wrong. It is chemistry - and an important part of chemistry is the classification of substances as acids and bases. All fruits are acidic and to a lesser extent so are vegetables. In apples for example, there are at least 20 organic acids, a small but significant portion of 130 different flavour and odour components so far identified and these acids themselves are metabolised in part to other flavour and odour components as they ripen. These aromas in turn have evolved to attract pollinating agents such as insects, birds and small mammals and to contribute to seed distribution and so continue the food chain.
DNA and RNA, the genetic material of living organisms are acids called nucleic acids. It is said DNA makes RNA makes protein. Proteins are made from amino acids. Glutamic acid, an amino acid used to make MSG used in Asian cooking, is a flavour found in tomatoes and oranges. Ascorbic acid and folic acid are vitamins. Hydrochloric acid in our stomach is part of the digestion process, but cooking itself makes many foods more digestible and the acidity of the cooking medium modifies taste, colour and texture.
An acid taste is a sour taste. Acids are responsible for the sour taste. The word acid is derived from the Latin, acidus meaning sour. The word for acid in German is saüre as in saüerkraut or pickled cabbage which is made using lactic acid.
Acids are preservatives, discouraging microbial action. Our skin secretions are acidic. Vinegar is made from sour wine - in French translated as vin aigre, which as well as being a preservative contributes to the odour in foods such as fish and chips.
Bitterness is the taste associated with water soluble bases called alkalis and being able to perceive bitterness protects humans from plant poisons some of which in turn are natural plant insecticides such as nicotine. Many plants and some animals use these bases called alkaloids, which are poisonsto deter predators. This destroys the assumption that 'natural chemicals' are safe, even if present in organically grown foods. Chemical warfare began in the plant kingdom!
Bases are the complementary group to acids. They neutralise acids forming salts.
The classification of acids and bases by physical and chemical properties
It was Robert Boyle who in the seventeenth century first brought order to the classification of acids and bases. He was only familiar with water-soluble bases or alkalis.
Acids - sour taste
- corrosive
- change litmus (a dye extracted from lichens) from blue to red.
- become less acidic when combined with alkalis
Alkalis - feel slippery
- change litmus from red to blue
- become less alkaline when combined with acid.
The classification of acids bases and salts by chemical composition
Oxygen, named by Antoine Lavoisier, the so-called 'Father of Modern Chemistry,' means 'acid generator' -because Lavoisier believed oxygen is the essential element in acids. He also called binary compounds of oxygen, oxides,
As you know already this is only true of non-metal oxides.
CO2, SO2, SO3, and NO2 all dissolve in water producing acids - H2CO3, (carbonic acid), H2SO3, (sulfurous acid), H2SO4, (sulfuric acid) and HNO3, (nitric acid) respectively. The metal oxides CaO and MgO produce alkaline solutions. In fact, all metal oxides are basic, and all acidic oxides are non metal oxides.
Just to confuse matters, some non-metal oxides are neither acidic nor basic. Carbon monoxide and nitric oxide, NO fall into this category and so does water. Have you heard the statement, all buses are vehicles, but not all vehicles are buses? This may help you to make sense of the statement, 'All acidic oxides are non-metal oxides, but not all non-metal oxides are acidic.'
It was Sir Humphry Davy early in the 19th century who noted that hydrochloric acid, HCl does not contain oxygen and soon after HI, HF, and HBr, all acids, were discovered as well.
In 1838 Justus von Liebig noted that acids contain at least one hydrogen atom. Thus a salt is a compound where at least one hydrogen in an acid has been replaced by a metal ion.
Bases are recognised as metal oxides, metal hydroxides, metal carbonates, metal bicarbonates (also called hydrogen carbonates), and ammonia.
Today's classification of acids and alkalis by physical and chemical properties has been expanded to include the following.
Properties of aqueous acids |
Properties of aqueous alkalis |
Sour taste
|
Bitter taste; slippery feel, dissolve fats.
|
They change the colour of indicators
Blue litmus turns red. Green Universal indicator turns red, (orange or yellow). Yellow or orange methyl orange turns red. Red phenol phthalein turns colourless.
|
They change the colour of indicators Red litmus turns blue. Green Universal indicator turns blue, indigo or violet. Red or orange methyl orange turns yellow. Colourless phenol phthalein turns pink.
|
They are corrosive
Active metals are eaten away (dissolve) in acids releasing hydrogen gas. Strong acids eat flesh. Mg + H2SO4→ MgSO4 + H2
|
Active metals do not react with alkalis. The metalloid aluminium is eaten away (dissolves) in alkalis producing hydrogen gas. |
They neutralise bases
Acid + metal oxide or metal hydroxide → salt + water H2SO4 + CuO → CuSO4 + H2O HCl + NaOH → NaCl + H2O
|
They neutralise acids
Alkali + acid or acidic oxide → salt + water KOH + HNO3 → KNO3 + H2O Ca(OH)2 + SO3→ CaSO4 + H2O
|
They neutralise carbonates and hydrogen carbonates (also bases), producing carbon dioxide gas.
Acid + metal carbonate or hydrogen carbonate → salt + water + carbon dioxide H2SO4 + CaCO3→CaSO4 + H2O + CO2 HCl + NaHCO3 → NaCl + H2O + CO2
|
Carbonates and hydrogen carbonates (also bases), neutralise acids producing carbon dioxide gas.
Solid carbonates such as limestone and marble dissolve in dilute aqueous acid and effervescence (fizz) is seen. (Equations in the left hand column)
|
Acids neutralise ammonia gas and aqueous ammonia
Acid + ammonia gas → ammonium salt Acid + ammonium hydroxide → ammonium salt + water HCl + NH3→ NH4Cl HNO3 + NH4OH → NH4NO3 + H2O
|
|
Acid and alkaline solutions are also electrolytes. Alkalis except for ammonia are ionic compounds. Acids and ammonia although covalent, react with water producing ions. Ions carry the current in electrolytes.
Tests for acids and alkalis
This classification of acids and alkalis also provides a means of identifying the presence of these compounds.
Tests for Acids.
1 All aqueous acids change the colour of the same indicators in the same way.
2 They all react with carbonates and bicarbonates producing bubbles of colourless gas.
3 They dissolve active metals such as magnesium producing a colourless gas, which can be identified as
hydrogen as it produces small explosions with the heat from a lighted match. (The pop test)
4 Water insoluble acidic oxides, like SiO2 dissolve in hot, concentrated alkali.
The use of taste to identify an acid is forbidden in a school laboratory.
Tests for Bases
1 Dilute alkalis have a slippery feel because they react with greasy secretions on the skin forming soap.
2 All aqueous alkalis change the colour of the same indicators in the same way.
3 Water insoluble bases such as copper(II) oxide dissolve when heated with hot acid.
4 Water soluble and water insoluble carbonates such as marble and chalk, (CaCO3) fizz and dissolve in
dilute aqueous acid.
5 Ammonia gas is easily identified by its pungent smell and its fumes turn moist red litmus paper blue.
Strong and weak acids and alkalis
The strong acids seen in a school laboratory are hydrochloric acid HCl, sulfuric acid H2SO4 and nitric acid HNO3 all of which occur naturally but are also referred to as mineral acids because they can be derived from minerals. Sulfuric acid, for example is made industrially on an enormous scale starting from sulfur.
Phosphoric acid, H3PO4 weaker than the other three is also classified as a mineral acid.
Strong acids react readily.
One of the weakest acids, carbonic acid, H2CO3 is found in clean rainwater and carbonated drinks. It turns green universal indicator from green to orange and barely gives the sensation of sourness in carbonated drinks. Heating decomposes aqueous carbonic acid to carbon dioxide and water and so it cannot be isolated pure.
Organic acids are weak acids and include:
Acetic acid (also called ethanoic acid) CH3CO2H, which gives the sourness to vinegar,
Tartaric acid, found in grapes,
Oxalic acid found in rhubarb and soursobs,
Citric acid and malic acids found in many fruits,
Lactic acid found in sour milk, muscle and fermented cheese,
Prussic acid, HCN used in chemical warfare by the bombardier beetle.
Butanoic acid found in Parmesan cheese and vomit.
Weak acids react less readily as your teacher will demonstrate - but they may have a strong smell!
The strongest alkali you will encounter in the school laboratory is sodium hydroxide, NaOH also called caustic soda. It is a piranha in the chemical world. It eats flesh. In significant quantities it will boil water when added to it. It absorbs water spontaneously from the atmosphere forming sticky pellets and eventually a solution. Solids with this property are said to be deliquescent. Alkalis have a slippery feel because they react with fats forming soap. The fatty tissues in your hand are turned to soap if dipped into caustic soda and your hand may never recover. Always wear rubber gloves and eye protection when cleaning a greasy oven or clearing a grease-blocked drain with sodium hydroxide.
Ammonia, bicarbonates and soluble carbonates are weak alkalis.
Strong alkalis react readily.
Weak alkalis react less readily.
Dilute and concentrated
Hydrogen chloride, HCl, is a gas, nitric, sulfuric and acetic acids are liquids and citric, oxalic and tartaric acids are solids. Either a little or a lot can be dissolved in water. Sulfuric acid is available commercially as 98% H2SO4 and is thus a strong, concentrated acid. 98% Acetic acid is a weak concentrated acid. Whether there is a lot or a little water in acid or alkaline solutions does not change their classification as strong or weak. Even 0.1% sulfuric acid is still a strong acid despite the fact it is very dilute. You are not eating a strong acid however unripe the rhubarb. The weak organic acids present are simply more concentrated in unripe fruits.
Explaining the common properties of acids and bases - theory
The Arrhenius theory - the classification of acids and alkalis by a common species in solution
Svante Arrhenius a Nobel Laureate in chemistry proposed that acids ionise in water - always producing the hydrogen ion.
HCl(g) + aq → H+(aq) + Cl – (aq)
This hydrogen ion a common feature of all acid solutions thus produces all the common properties - sourness, effervescence with active metals, carbonates and bicarbonates and the change of colour of indicators. This also explains why acids are electrolytes. Ions carry the current!
He also explained why strong acids like HCl are good conductors. Strong acids ionise completely in water.
CH3COOH(l) + aq - - - - -> H+(aq) + CH3COO – (aq)
Weak acids like acetic acid are poor electrolytes. They ionise only partially producing a low concentration of ions. At room temperature, only about 3 in every one thousand acetic acid molecules ionises.
Arrhenius defined a base as any substance that dissolves in water producing the hydroxide ion. This includes the dissociation of water-soluble hydroxides,
NaOH(s) + aq → Na+(aq) + OH – (aq)
the reaction of water soluble oxides such as sodium oxide and calcium oxide with water,
Na2O(s) + H2O(l)→ 2Na+(aq) + 2OH – (aq) ,
CaO(s) + H2O(l) → Ca++(aq) + 2OH – (aq)
and the ionisation of ammonia:
NH3(g) + H2O(l)- - - - - - > NH4+(aq) + OH –aq)
Strong bases like sodium hydroxides conduct well in solution. Ammonia, which is a weak base as it ionises only partially in water, conducts poorly.
The Arrhenius theory has several drawbacks, one being that it limits acid base chemistry to reactions in water. Water insoluble acids and bases are excluded. A theory was needed which was independent of solvent so as to include reactions such as the reaction of ammonia gas with hydrogen chloride gas producing the salt ammonium chloride.
NH3(g) + HCl(g) → NH4Cl(s)
Water soluble bases are now called alkalis
The Lowry Brǿnsted theory
In 1923, an Englishman Thomas Lowry, and a Dane Nicolaus Brǿnsted, independently, and almost instantaneously proposed a new theory which now bears their names.
An acid is a proton donor
In fact an acid is only a potential proton donor unless there is another substance present which will accept them. This can be explained by using the reaction of hydrogen chloride gas with water. This gives an acidic solution, hydrochloric acid.
Bases are proton acceptors
Neither hydrogen chloride nor water conducts because there are no ions present. When hydrogen chloride dissolves in water, hydrogen transfer occurs producing ions. Hydrochloric acid forms which does conduct.
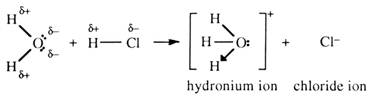
The hydrogen ion donated by hydrogen chloride attaches itself to the accepting molecule water to form the hydronium ion, H3O+. The hydronium ion is also written as H+(aq). The reaction occurs because of the polar bonds present in both molecules. Without polarity there would be no reaction. The δ+ attracts the δ –. A non-bonding pair on the oxygen atom forms a covalent bond with the proton. This bond is sometimes called a coordinate covalent bond since the oxygen supplies both bonding electrons. Notice here that since water is the proton acceptor, it is acting as a base.
A chemical change that produces ions, is called ionisation.
The proton transfer that occurs from an acid to a base is called protolysis
A protolysis reaction without water is when ammonia gas, NH3(g) meets hydrogen chloride gas. There is a rapid reaction and white clouds of ammonium chloride, NH4Cl(s) form.
HCl(g) + NH3(g)→ NH4Cl(s)
The formation of ammonium chloride involves two processes:
● the donation of protons
HCl → H+ + Cl –
● the acceptance of protons
NH3 + H+ → NH4+
The ammonia molecule has a non-bonding pair on the nitrogen atom which forms a bond with the proton.
Overall the reaction is: NH3(g) + HCl(g) → NH4+ + Cl – |
another co-ordinate covalent bond |
The ions attract each other to make the solid ammonium chloride.
When ammonia ionises in water, the non-bonding pair on ammonia accepts a proton from water.
H2O + NH3 → NH4+ + OH –
In this protolysis reaction water is acting as the acid.
Amphiprotic and amphoteric substances
There are some substances that can accept protons and donate protons. Water is one of these and is said to be amphiprotic. It can react with itself producing hydronium ions and hydroxide ions in equal numbers but since it is both an extremely weak acid and an extremely weak base the concentration of ions is very, very small.
H2O + H2O - - - - -> H3O+ + OH –
What is the difference between an amphoteric substance and an amphiprotic substance? Not much, but it is important that you know the difference. Al2O3 is amphoteric. It can react with both hot acid and hot alkali but it is not amphiprotic because it cannot donate a proton.
Conjugate acids and bases
When an acid loses a proton to form a base, the only difference in its formula is H+, such as with HCl and Cl –. The Cl – formed by loss of a proton is a potential proton acceptor and could reform HCl. The HCl and Cl – are referred to as a conjugate acid-base pair and this is a complicated way of stating a very simple relationship between them. Other conjugate acid base pairs are HNO3 / NO3–, H2SO4 / HSO4– and NH4+ / NH3. The nitrate ion, NO3– the hydrogen sulfate ion, HSO4– and ammonia are all potential proton acceptors to form their so-called conjugate acids. Notice that the Lowry-Brǿnsted theory includes ions as well as neutral molecules as potential proton donors or acceptors.
In any acid base-reaction, there are two conjugate acid-base pairs.
Consider the reactions below:
HNO3 + H2O → NO3 – + H3O+
acid 1 base 2 base 1 acid 2
The acid - base pairs are identified by numbers. One acid-base pair is HNO3 / NO3– - labelled here as acid 1 and base 1; the other is H2O / H3O+ labelled base 2 and acid 2.
Acid and base strength
What is a strong acid or base in terms of the proton theory? Strong acids donate protons readily whereas weak acids do not donate protons easily. HCl is a strong acid. In water its donation of protons is complete.
HCl + H2O → H3O+ + Cl –
A strong base readily accepts protons. The hydroxide ion is a strong base as it readily accepts protons.
H3O+ + OH – → H2O + H2O
Because HCl(g) is a strong acid it follows that its conjugate base must be very weak. If Cl– accepted protons readily, it would reform HCl and so HCl would not be donating all of its protons. Thus for HCl:
HCl → H+ + Cl –
strong acid weak base
The conjugate acid of a strong base must be weak. Thus for the hydroxide ion:
H+ + OH – → H2O
strong base weak acid
Polyprotic acids
Sulfuric acid is diprotic, meaning it has two transferable protons as the equations below show.
H2SO4 + H2O → HSO4– + H3O+
acid 1 base 2 base 1 acid 2
HSO4– + H2O → SO42 – + H3O+
acid 1 base 2 base 1 acid 2
Note that here the hydrogen sulfate ion in the first ionisation step is a conjugate base but in the second step it is a proton donor and so an acid. The hydrogen sulfate ion is amphiprotic!
Polyprotic acids may be weak or strong. Sulfuric acid is a strong acid but carbonic acid which like sulfuric acid is diprotic, is extremely weak. Citric acid which is triprotic, is also very weak.
Measuring the concentration of H3O+ and OH – in aqueous solution - the pH scale
In cooking, in swimming pools, in growing plants, in blood and in all the vital organs of the body, the right concentration of H3O+ and OH – is crucial for proper functioning - even though these concentrations are so very, very small. No living organism has concentrated sulfuric acid in its tissues! Thus a measure of acidity or alkalinity is essential and a simple pH scale between the values of 1 and 14 shows how.
1 2 3 4 5 6 7 8 9 10 11 12 13 14
<------------ACIDIC---------------------------NEUTRAL---------------------ALKALINE------------------------>
|
The pH of some common substances |
|
The H in pH stands for hydrogen, so it must always be a |
human gastric juice |
1-3 - 3.0 |
capital H! |
lemon juice |
2.1 |
Water has a tiny but equal number of H3O+ and OH – ions so |
orange juice |
3.0 |
water is neutral with a pH of 7 |
yoghurt |
4.5 |
In acid solutions there is a surplus of H3O+ ions. Any aqueous |
black coffee |
5.0 |
solution with a pH of less than 7 is acidic. |
milk |
6.9 |
In an alkaline solution there is a surplus of OH – ions. An aqueous |
egg white |
7.6 - 9.5 |
solution with a pH of greater than 7 is alkaline. |
baking soda in water |
8.4 |
pH can only be measured in aqueous solutions. |
household ammonia |
11.9 |
The greater the hydrogen ion concentration, the lower the pH.
A chemist measures pH using ● indicators
● pH paper
● a pH meter
An indicator shows one colour in an acid and another colour in an alkaline solution. On mixing a number of indicators together the chemist has a means of comparing the hydrogen ion concentration of acids and the hydroxide concentration of alkalis. These indicator mixtures are called universal indicators which change colour over a wide range of acidity and alkalinity. In universal indicators each different colour indicates a different pH in a range of 3 to 11.
The colours are those in the rainbow - red, orange, yellow, green, blue, indigo and violet.
●. Green shows a pH of 7
● Red, orange & yellow indicate acidity and red is marked pH 3
but red indicates a pH of 3 or less.
● Blue, indigo and violet show that the solution is alkaline.
Violet marked pH 11, indicates a pH of 11 or more.
Universal indicator only gives pH in whole numbers.
Calculating pH
By convention, the concentration of H+(aq) or H3O+ in mol L-1can be written as [H+]
This gives a mathematical definition of pH.
pH = – log10[H+].
Using a log scale, pH is given in bigger numbers but a small difference in pH is a big difference in
[H+] as the table below shows.
[H+] |
[H+] (scientific notation) |
pH = – log10[H+] |
|
0.1 |
1 x 10-1 |
1 |
1,000,000X more acidic than pH 7 |
0.01 |
1 x 10-2 |
2 |
|
0.001 |
1 x 10-3 |
3 |
|
0.0001 |
1 x 10-4 |
4 |
|
0.00001 |
1 x 10-5 |
5 |
10X more acidic than pH 6 |
0.000001 |
1 x 10-6 |
6 |
|
0.0000001 |
1 x 10-7 |
7 |
|
0.00000001 |
1 x 10-8 |
8 |
|
0.0000000001 |
1 x 10-10 |
10 |
100X more alkaline than pH 8 |
0.0.000000000001 |
1 x 10-12 |
12 |
|
0.00000000000001 |
1 x 10-14 |
14 |
10,000X more alkaline than pH 8 |
Writing equations for acid base reactions
Formulae equations show which acid, which base and which salt is formed when they neutralise each other.
Salts derive their names from the acid and base neutralised to form them - so
The salts made from hydrochloric acid, HCl are chlorides.
The salts made from nitric acid HNO3 are nitrates.
The salts made from sulfuric acid H2SO4 are sulfates.
The salts made from acetic acid, CH3CO2H are acetates (or ethanoates).
The salts made from phosphoric acid H3PO4 are phosphates
The salts made from sulfurous acid H2SO3 are sulfites.
The cations are metal ions or ammonium ions, giving salts such as potassium nitrate, KNO3 and ammonium chloride, NH4Cl.
Common laboratory alkalis are:
Sodium hydroxide, NaOH, common name caustic soda - a white solid which is colourless in solution.
Potassium hydroxide, KOH, common name caustic potash - a white solid which colourless in solution.
Calcium hydroxide, Ca(OH)2, common name, slaked lime - a white solid forming a colourless solution called limewater.
Ammonia solution, NH3(aq) It is a colourless gas forming a colourless solution which has a powerful and pungent odour.
Uses of Alkalis
The uses of alkalis are many and lime, referring to both calcium oxide and calcium hydroxide, is one of the top five 'heavy industrial chemicals produced world wide. Lime is one of the oldest chemicals known to humankind and has been used for thousands of years in agriculture to improve crop yields by raising the pH of soils. It is prepared by heating limestone, CaCO3 to liberate carbon dioxide.
CaCO3 → CaO + CO2
Calcium oxide is produced by this reaction in blast furnaces. Lime is added to blast furnaces together with coke (the fuel), and iron ore. Calcium oxide reacts with the solid acidic oxide, SiO2 and so separates this impurity as a separate layer called slag from the molten iron. The slag is less dense than iron and floats on top. The blast furnace is not only a reaction vessel but an enormous separating funnel. The molten iron can be tapped off. Iron has more applications than any other metal and accounts for over 90% of all metal refined. Acid base chemistry has an important part to play in its production.
CaO + SiO2 → CaSiO3 - calcium silicate slag.
Calcium oxide called quicklime because of its rapid and exothermic reaction with water, forms calcium hydroxide, which in solution is called limewater. Wooden sailing ships carrying quicklime sometimes caught fire when they leaked water into the hold.
CaO + H2O →Ca(OH)2
Lime is used to remove sulfur dioxide in a process called 'flue gas desulfurisation.' This pollutant is produced in fossil fuel power stations and unless removed by this 'wet scrubbing' process is widely dispersed in the atmosphere after emissions from tall smoke stacks. Neutralisation to the rescue again!
Ca(OH)2 + SO2 → CaSO4 + H2O
Both sodium hydroxide and ammonia are in the top 10 industrial chemicals. Ammonium salts are used in an enormous scale in agriculture as fertilisers, although the use of ammonium nitrate is now restricted because of its use by terrorists as a readily available explosive.
Lithium oxide is used to scrub air free of carbon dioxide in manned space ships and submarines. This neutralisation helps to ensure that the astronauts and submariners do not suffocate.
2LiOH + CO2 → Li2CO3 + H2O
Sodium hydroxide which is deliquescent absorbs carbon dioxide spontaneously from the atmosphere and so converts itself to sodium carbonate in yet another neutralisation reaction of an acidic oxide with an alkali.
Writing ionic equations
Ionic equations are general equations showing why all acids have common properties. The hydronium ion, H+(aq) is the common reactant in each one.
Acids neutralise metal hydroxides
Acid + metal hydroxide → salt + water
HCl + NaOH → NaCl + H2O
The strong acid ionises completely and NaOH is dissociated as it is in aqueous solution.
In ionic form, the equation can be rewritten:
H+ + Cl – + Na+ + OH – → Na+ + Cl – + H2O
The sodium ions and chloride ions, which appear on both sides of the equation, are called spectator ions.
They do not come together unless evaporation occurs.
The actual reaction is thus:
H+ + OH – → H2O
The ionic equation for any acid reacting with any metal hydroxide
or H3O+ + OH – → 2H2O
Acids neutralise metal oxides
Acid + metal oxide → salt + water
2HNO3 + CaO → Ca(NO3)2 + H2O
In ionic form the equation can be rewritten
2H+ + 2NO3– + Ca2+ + O2 –→ Ca2+ + 2NO3– + H2O
The calcium ions and nitrate ions, which appear on both sides of the equation, are called spectator ions.
They do not come together unless evaporation occurs.
The actual reaction is thus:
2H+ + O2 – → H2O
The ionic equation for any acid reacting with any metal oxide
Acids neutralise carbonates and bicarbonates.
acid + carbonate → salt + water + carbon dioxide
H2SO4 + Na2CO3→ Na2SO4 + H2O + CO2
In ionic form, the equation can be rewritten:
2H+ + SO42 – + 2Na+ + CO32 – → 2Na+ + SO42 – + H2O + CO2
The sodium ions and sulfate ions, which appear on both sides of the equation, are called spectator ions.
The actual reaction is thus:
2H+ + CO32 – → H2O + CO2
The ionic equation for any acidreacting with any carbonate
acid + hydrogen carbonate → salt + water + carbon dioxide
2HNO3 + Mg(HCO3)2→ Mg(NO3)2 + 2H2O + 2CO2
The actual reaction is thus:
H+ + HCO3– → H2O + CO2
The ionic equation for any acid reacting with any bicarbonate
Acids corrode active metals
acid + active metal → salt + hydrogen gas
2HCl + Ca → CaCl2 + H2
In ionic form, the equation can be rewritten
2H+ + 2Cl – + Ca → Ca2+ + 2Cl – + H2
The chloride ions which appear on both sides of the equation are called spectator ions.
The actual reaction is thus:
2H+ + Ca → Ca2+ + H2
The ionic equation for any acid reacting with calcium
or for any active divalent metal M:
2H+ + M → M2+ + H2
Acidic oxides in the atmosphere Bases in the Earth's crust
Acidic oxides are covalent oxides and are present in our atmosphere.
Carbon dioxide is moderately soluble in water and forms carbonic acid. Pure rainwater is very dilute carbonic acid.
H2O + CO2 → H2CO3
Nitric oxide forms from atmospheric nitrogen and oxygen when energy is supplied in the form of lightning.
N2 + O2 → 2NO
This in turn oxidises to nitrogen dioxide.
2NO + O2 → 2NO2
Nitrogen dioxide is an acidic oxide and dissolves in water forming nitric acid.
3NO2 + H2O → 2HNO3 + NO
These gaseous acidic oxides together with sulfuric acid and sulfuric acids formed from their acidic oxides
in the atmosphere, react with the ionic bases in the Earth's crust such as Fe2O3, MgCO3 and CaCO3 as well as active metals like iron, zinc and magnesium. Globally acid-base chemistry occurs on an enormous scale and is boosted by acidic oxide pollutants as you will learn in the topic 'Acid Rain.'
Exercises
SKILL DRILLS Complete and balance the equations below.
(a) Name the acids formed. (b) Write equations to show the ionisation of
these acids in water.
1. CO2 + H2O → 2. SO2 + H2O → 3. NO2 + H2O → 4. SO3 + H2O →
|
|
(c) Write complete, balanced, formulae equations for each of the following:
1. Sulfuric acid + sodium oxide 2. Sulfuric acid + copper(II) oxide 3. Sulfuric acid + iron(III) oxide 4. Sulfuric acid + aluminium oxide 5. Sulfuric acid + sodium hydroxide 6. Sulfuric acid + sodium carbonate 7. Sulfuric acid + sodium bicarbonate 8. Sulfuric acid + ammonia 9. Sulfuric acid + water 10. Sulfuric acid + iron 11. Sulfuric acid + zinc 12. Sulfuric acid + magnesium
|
13. Hydrochloric acid + aluminium 14. Nitric acid + sodium oxide 15. Nitric acid + copper(II) oxide 16. Nitric acid + iron(III) oxide 17. Nitric acid + aluminium oxide 18. Nitric acid + sodium hydroxide 19. Nitric acid + sodium carbonate 20. Nitric acid + sodium bicarbonate 21. Nitric acid + ammonia 22. Nitric acid + water 23. Nitric acid + magnesium 24. Ethanoic acid + ammonia
|
(d) Write balanced ionic equations for each of the following:
1. Sulfuric acid + sodium oxide 2. Sulfuric acid + sodium carbonate 3. Sulfuric acid + calcium carbonate 4. Nitric acid + sodium bicarbonate 5. Nitric acid + sodium hydroxide |
6. Nitric acid + copper(II) oxide 7. Nitric acid + magnesium 8. Sulfuric acid + magnesium 9. Ethanoic acid + magnesium 10. Sulfurous acid + magnesium carbonate
|
|
|
Source: http://www.usc.adelaide.edu.au/local/transitionlectures/chemistry/acids_bases_salts.doc
Web site: http://www.usc.adelaide.edu.au
Author : not indicated on the source document of the above text
Google key word : Acids bases and salts file type : doc
If you are the author of the text above and you not agree to share your knowledge for teaching, research, scholarship (for fair use as indicated in the United States copyrigh low) please send us an e-mail and we will remove your text quickly.
Acid-Base Theories
KEY CONCEPTS
- What are the properties of acids and bases?
- How did Arrhenius define an acid and a base?
- What distinguishes an acid from a base in the Brönsted-Lowry theory?
- How did Lewis define an acid and a base?
--Many everyday materials contain acids or bases
--Acids
1. taste sour
2. change the color of an acid-base indicator
3. are electrolytes
--Bases
1. taste bitter
2. feel slippery
3. change the color of an acid-base indicator
4. are electrolytes
--An ARRHENIUS ACID is a hydrogen-containing compound that ionizes in aqueous solution to produce hydrogen ions (H+)
--An ARRHENIUS BASE is a compound that produces hydroxide ions (OH-) in aqueous solution
--A MONOPROTIC ACID is one that contains only one ionizable hydrogen
Ex. HCl, HNO3
--A POLYPROTIC ACID is one that contains 2 or more ionizable hydrogens
Ex. H2SO4- DIPROTIC, H3PO4- TRIPROTIC
--Note that only hydrogens in very polar bonds are ionizable
Ex. CH3COOH
--Random facts about bases:
1. NaOH (lye) and KOH (found in ashes) are very soluble bases and extremely caustic
2. Ca(OH)2 (lime) and Mg(OH)2 (in milk of magnesia) are not very soluble
--What do the formulas of the bases above have in common?
--A BRÖNSTED-LOWRY ACID is a proton/hydrogen ion (H+) donor
--A BRÖNSTED-LOWRY BASE is a proton/hydrogen ion (H+) acceptor
Ex. NH3(aq) + H2O(l) £ NH4+(aq) + OH-(aq)
Which reactant is a proton donor? Proton acceptor?
B-L base? B-L acid?
--A CONJUGATE ACID is what the base becomes after it accepts a proton
--A CONJUGATE BASE is what the acid becomes after it donates a proton
Ex. What is the conjugate acid and base in the following reactions
HCl(g) + H2O(l) £ H3O+(aq) + Cl-(aq)
NH3(aq) + H2O(l) £ NH4+(aq) + OH-(aq)
Ex. What is the conjugate acid of each of the following:
NH3, H2O, OH-, HCO3-, CO32-, NH2-
Ex. What is the conjugate base of each of the following:
NH3, H2O, HCO3-, H2CO3, H2SO4, HSO4-
--A HYDRONIUM ION (H3O+) is the ion that results when a water molecule gains a hydrogen ion
--An AMPHOTERIC substance is one that can behave like an acid or base
--Which substances in the examples above are amphoteric?
--A LEWIS ACID is an electron pair acceptor; a LEWIS BASE is an electron pair donor
Ex. H+ + OH-
--An ACIDIC ANHYDRIDE is a nonmetallic oxide that, when dissolved in water, produces an acid
Ex. CO2(g) + H2O(l) £ H2CO3(aq)
--A BASIC ANHYDRIDE is a metallic oxide that, when dissolved in water, produces a base
Ex. Na2O(s) + H2O(l) ß 2NaOH(aq)
--Are the following acidic or basic anhydrides?
CaO, SO2, NO2, K2O, BaO
SEE 19.1 SECTION ASSESSMENT, P. 593
Section 19.2-Hydrogen Ions and Acidity
KEY CONCEPTS
- How are [H+] and [OH-] related in an aqueous solution?
- How is the hydrogen-ion concentration used to classify a solution as neutral, acidic, or basic?
- What is the most important characteristic of an acid-base indicator?
--Water is amphoteric (meaning what?) and can undergo SELF (AUTO)-IONIZATION
H2O(l) + H2O(l) £ H3O+(aq) + OH-(aq)
or
H2O(l) £ H+(aq) + OH-(aq)
--In pure water, [H3O+] = [OH-] = 1 X 10-7 M
--Since an equilibrium exists between water molecules, hydronium ions, and hydroxide ions
Keq = [H3O+][OH-]/[H2O]2
As [H2O] is much larger than [H3O+] and [OH-] and is essentially constant, one can move [H2O]2 to the other side of the equation to obtain
[H2O]2 Keq = [H3O+][OH-]
--The ION PRODUCT CONSTANT FOR WATER (Kw) is the product of the concentrations of H3O+ (or H+) and OH-
Kw = [H3O+][OH-] = (1 X 10-7 M)2 = 1 X 10-14
--What happens to the concentration of H3O+ (H+) if an acid is added to water? OH- concentration? If a base is added?
Ex. SEE SAMPLE PROBLEM 19.1, PRACTICE PROBLEMS 9, 10, P. 596
--The pH of a solution is the negative logarithm of [H3O+] (or [H+])
pH = -log[H3O+]
Ex. What is the pH of a neutral solution?
Ex. What is the pH of a solution with [H+] = 6 X 10-10 M
Ex. What is the [H3O+] ([H+]) of a solution with
pH = 4.8?
--To summarize
1. An ACIDIC SOLUTION is one in which
[H3O+] > [OH-]
pH < 7
[H3O+] > 1 X 10-7M
2. A BASIC (ALKALINE) SOLUTION is one in which
[H3O+] < [OH-]
pH > 7
[H3O+] < 1 X 10-7M
3. A NEUTRAL SOLUTION is one in which
[H3O+] = [OH-] = 1 X 10-7M
pH = 7
--pOH is the negative logarithm of the [OH-]
pOH = -log[OH-]
--If one takes the negative logarithm of both sides of the Kw equation- Kw = [H3O+][OH-] = 1 X 10-14
the result is another useful equation
pH + pOH = 14
--Given one of the following
[H3O+] [OH-] pH pOH
one should be able to calculate the other three values for an aqueous solution
Ex. A solution has a [H3O+] = 4.2 X 10-10 M. What is the [OH-], pH, and pOH?
Ex. A solution has a pH = 9.2. What is the [H3O+], pOH, and [OH-]?
Ex. A solution has a [OH-] = 3.5 X 10-3 M. What is the [H3O+], pH, and pOH?
Ex. A solution has a pOH = 4.5. What is the [OH-], pH, and [H3O+]?
--An INDICATOR (HIn) is a weak acid or base that undergoes a color change at a specific pH when the molecule gains or loses a proton
HIn £ H+ + In-
SEE FIG. 19.12, P. 602
Ex. Universal indicator, pH paper, red/blue litmus paper
--A pH meter is an electrical instrument used to make rapid, accurate pH measurements
SEE FIG. 19.15, P. 603
SEE 19.2 SECTION ASSESSMENT, P. 604
Section 19.3-Strengths of Acids and Bases
KEY CONCEPTS
- How does the value of an acid dissociation constant relate to the strength of an acid?
- How can you calculate an acid dissociation constant (Ka) of a weak acid?
--A STRONG ACID is one that ionizes completely in aqueous solution
Ex. HCl(g) + H2O(l) ß H3O+(aq) + Cl-(aq)
--A WEAK ACID is one that only partially ionizes in aqueous solution
Ex. CH3COOH(aq) + H2O(l) £ CH3COO-(aq) + H3O+(aq)
--The strong binary acids are HCl, HBr, HI
--A rule-of-thumb for strong ternary acids is
“If the number of oxygens exceeds the number of hydrogens by two or more, the acid is strong”
Ex. H2SO4-strong H2SO3-weak
Ex. What is the concentration of H3O+ in a 0.3 M solution of HNO3? What is the pH?
Ex. What is the pH of a 1.3 M solution of HCl?
--Since an equilibrium exists between a weak acid and its conjugate base, an equilibrium expression can be written; for a weak acid HA
HA(aq) + H2O(l) £ A-(aq) + H3O+(aq)
Keq = [A-][H3O+] / [HA][H2O]
[H2O] is essentially a constant, and can be moved to the other side of the equation to form a new constant- the ACID IONIZATION (DISSOCIATION) CONSTANT (Ka)
Ka = [A-][H3O+] / [HA]
--The larger Ka is for an acid, the stronger the acid
SEE TABLE 19.7, P. 607
--Write the Ka expression for each of the following
H2SO4, HSO4-, H2CO3, HCO3-, CH3COOH
--Note that the conjugate base of a polyprotic acid is a weaker acid than the original acid
Ex. H3PO4
--A STRONG BASE is one that dissociates completely in aqueous solution
--All soluble metallic hydroxides are strong bases, in particular Group 1 and heavy Group 2 hydroxides
--A WEAK BASE is one that reacts slightly with water to produce OH-
Ex. NH3(aq) + H2O(l) £ NH4+(aq) + OH-(aq)
--Note that the reaction in the example above lies to the left at equilibrium
Ex. What is the [OH-] of a 0.75 M NaOH solution? What is the pH?
--The BASE DISSOCIATION CONSTANT (Kb) for a weak base is analogous to Ka for a weak acid; for the weak base B
B + H2O £ HB+ + OH-
Kb = [HB+][OH-] / [B]
--Write the Kb expression for each of the following
NH3, NH2-, SO42-
--Is there a difference between concentration and strength of an acid/base? Can one have a weak acid/base solution with a high concentration?
--The Ka for a weak acid or Kb for a weak base can be calculated if one knows the initial concentration of the acid/base and the pH
Ex. SEE SAMPLE PROBLEM 19.5, P. 610
SEE 19.3 SECTION ASSESSMENT
Section 19.4-Neutralization Reactions
KEY CONCEPTS
- What are the products of the reaction of an acid with a base?
- What occurs at the endpoint of a titration?
--A SALT is a compound consisting of the anion from an acid and a cation from a base
Ex. HCl + NaOH ß NaCl + HOH
--The reaction in the example above is a NEUTRALIZATION REACTION-a reaction between an acid and a base that produces a salt and water
--Acid-base reactions are called neutralization reactions because if the reaction involves a strong acid and a strong base, and they are in stoichiometrically equivalent amounts, the resulting solution will be neutral
Ex. What acid/base combination could form the following salts (cation from base, anion from acid):
KCl, NaNO3, CaCO3, KBr, (NH4)2SO4
--TITRATION is a method which uses a standard solution to measure the concentration of another solution
--A STANDARD SOLUTION is a solution of known concentration
--In a titration, the solutes in the standard and unknown solution react. The standard solution is added to a given volume of unknown solution until the reaction is complete and there are stoichiometrically equivalent amounts of the two solutes-called the ENDPOINT. From the volumetric data obtained, the concentration of the unknown solution can be calculated
--In a strong acid-strong base titration (a neutralization reaction), the endpoint is reached when the pH is 7 (solution of a strong acid-strong base salt).
--Generally a titration would proceed as follows:
1. A volume of unknown solution is measured and placed in an appropriate container (Erlenmeyer flask)
2. Indicator (or a pH meter probe) is added to the unknown solution. An indicator commonly used in student laboratories is phenolphthalein (see Fig. 19.12, p. 602)
3. A BURET-graduated instrument used to measure out small volumes of liquid-is filled with the standard solution and an initial volume reading taken
4. Standard solution from the buret is released into the unknown solution until the slightest color change is detected
5. A final standard solution volume reading is taken
6. Calculation of unknown solution concentration is performed
Ex. The following volumetric data were obtained from the titration of an unknown HCl solution with a 0.75 M NaOH solution:
Volume of HCl solution - 15.50 mL
Initial buret reading - 0.52 mL
Final buret reading - 27.87 mL
Calculate the concentration of the unknown solution.
Ex. If 23.4 cm3 of 0.551M NaOH is used to titrate 50.0 cm3 of HCl to the endpoint, what is the concentration of the HCl solution?
Ex. How many cm3 of 0.0947M NaOH are needed to neutralize 21.4 cm3 of 0.106M HCl?
Ex. How many cm3 of 0.200M KOH will completely neutralize 15.0 cm3 of 0.400M H2SO4?
Section 19.5-Salts in Solution
KEY CONCEPTS
- When is the solution of a salt acidic or basic?
- What are the components of a buffer?
--While salts formed from reactions of strong acids with strong bases produce neutral solutions, this is not the case for salts of weak acids or bases
--In SALT HYDROLYSIS, ions from a salt react with water to produce an excess of H3O+ (H+) or OH-, which changes the pH of the solution
--Consider salts formed from the following acid/base neutralization reactions
1. STRONG ACID + WEAK BASE
Ex. Al(NO3)3 (from Al(OH)3 and HNO3)
Al3+ reacts with water to re-form the weak base
Al3+ + 3HOH £ Al(OH)3 + 3H+
Since this reaction lies to the right, and produces excess H+ (H3O+), the pH is lowered and an acidic solution is the result.
Will NO3- react with water to re-form HNO3? Why or why not?
2. WEAK ACID + STRONG BASE
Ex. NaCH3COO (from CH3COOH + NaOH)
CH3COO- reacts with water to re-form the weak acid
CH3COO- + HOH £ CH3COOH + OH-
Since this reaction lies to the right, and produces excess OH-, the pH is raised and a basic solution is the result.
Will Na+ react with water to re-form NaOH? Why or why not?
--Why will the salt from a strong acid/base reaction produce a neutral solution?
--What type of solution will a weak base/weak acid salt produce?
--To summarize
strong acid + strong base salt – neutral solution
strong acid + weak base salt – acidic solution
weak acid + strong base salt – basic solution
weak acid + weak base salt - ?
Ex. Will the following salts produce an acidic, basic, or neutral solution?
NaBr, NaSO3, CaSO4, NH4Cl, NH4NO3, KClO2
--A BUFFER is a solution of
1. a weak acid and one of its salts
or
2. a weak base and one of its salts
that resists changes in pH when small amounts of acid or base are added
--In a buffer system, an equilibrium is established between the weak acid and its conjugate base or the weak base and its conjugate acid. These provide species for added acid or base to react with instead of reacting with water and changing pH
Ex. CH3COOH and NaCH3COO buffer
The species present in this buffer system are
CH3COOH Na+ CH3COO- H2O
An equilibrium between acid and conjugate base is established
CH3COOH + H2O £ CH3COO- + H3O+
If acid is added to the system, the proton is donated to the strongest base present-acetate
If base is added to the system, the strongest acid donates a proton-acetic acid
In either case, the concentration of H3O+ doesn’t change significantly, nor does the pH
--The BUFFER CAPACITY of a buffer system is the amount of acid or base that can be added to a buffer solution before a significant change in pH occurs
--What would determine buffer capacity of a buffered system?
Source : http://www.duplinschools.net/22032033115139260/lib/22032033115139260/_files/Chemistry-Notes_Chapter_19_(Acids,_Bases,_and_Salts).doc
Web site link: http://www.duplinschools.net/
Author : not indicated on the source document of the above text
Google key word : Acids bases and salts file type : doc
If you are the author of the text above and you not agree to share your knowledge for teaching, research, scholarship (for fair use as indicated in the United States copyrigh low) please send us an e-mail and we will remove your text quickly.
Acids bases and salts
If you want to quickly find the pages about a particular topic as Acids bases and salts use the following search engine:
Chemistry
Acids bases and salts
Please visit our home page
Larapedia.com Terms of service and privacy page