Industrial chemistry and some important covalent
Industrial chemistry and some important covalent
The following texts are the property of their respective authors and we thank them for giving us the opportunity to share for free to students, teachers and users of the Web their texts will used only for illustrative educational and scientific purposes only.
The information of medicine and health contained in the site are of a general nature and purpose which is purely informative and for this reason may not replace in any case, the council of a doctor or a qualified entity legally to the profession.
![]()
Industrial chemistry and some important covalent
Chemistry
Heavy chemicals are the bulk chemicals used in industry and agriculture and are themselves produced from raw materials. These are accessed from the earth, sea and sky.
From the sky |
air |
needed as a coolant and to produce oxygen and nitrogen. |
From the sea |
water or steam |
a solvent, coolant and a source of other chemicals. (Na, Cl2) |
From the earth |
oil, natural gas |
The petrochemical industry provides polymers used to make plastics, fibres and elastomers such as polythene, polystyrene, nylon and PET. |
|
and minerals |
commonly oxides (Fe2O3, Al2O3), sulfides (NiS, ZnS, CuS) and carbonates, (CaCO3, MgCO3). |
Minerals are naturally occurring substances - metals or metal compounds - with a definite chemical composition and crystalline structure.
Coal, oil and natural gas supply most of the energy needed, with hydroelectric, geothermal, wind energy contributing, depending on locality. Energy efficiencies are being developed to conserve resources and reduce cost, particularly where electrical energy is needed.
EU and the US followed by Japan are the largest producers of chemicals, with BASF (Germany) and Dow Chemicals (US) in the number 1 and 2 positions respectively. Currently the world's chemical output is estimated at 3.7 trillion dollars US.
Sulfuric acid
Sulfuric acid, H2SO4, is the largest volume industrial chemical produced in the world. The consumption of sulfuric acid is often used to monitor a country's degree of industrialisation. It is a viscous, high density liquid, known in antiquity as oil of vitriol. It is sold as 98% (concentrated), 10% (dilute) or 29-32% in car batteries. |
O –H | O=S=O | O –H The valence bond structure of sulfuric acid The shape around the central sulfur atom is tetrahedral. |
65% sulfuric acid is used in fertiliser production, but it is also used to make dyes, paints, detergents, dyes, films, explosives and drugs.
It is formed naturally in some mining areas by the oxidation of sulfide minerals such as iron sulfide. The yellow areas on Jupiter's moon Europa found by NASA's spacecraft Galileo, have been identified as areas of high concentration of frozen sulfuric acid.
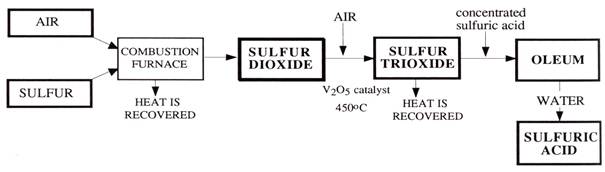
The Contact Process
Almost all sulfuric acid in the world today is made by the Contact Process first patented in 1831 and so called because in a key step, the reactants are in contact with the catalyst, vanadium pentoxide V2O5.
The steps are 1 burning sulfur in air to form sulfur dioxide
S + O2 → SO2
This reaction is exothermic and the heat given out is used to
(a) maintain the temperature of the process
(b) generate electricity to run the plant.
2 oxidising the sulfur dioxide to sulfur trioxide.
2SO2 + O2 → 2SO3
The reaction is slow and the V2O5 catalyst speeds up the rate of reaction, at a moderate
temperature of about 450oC.
Extra oxygen and a slight increase in pressure are used to ensure a high conversion.
3 The sulfur trioxide is dissolved in concentrated sulfuric acid to form another acid called
oleum., H2S2O7. Oleum means 'oil-like'.
SO3 + H2SO4 → H2S2O7
4 Conversion of oleum to sulfuric acid of the required concentration can occur on
site, saving on transport costs of water.
H2S2O7 + H2O → 2H2SO4
Another reason why water is not used to dissolve the sulfur trioxide directly to form
sulfuric acid is because sulfur trioxide would react with the water vapour above the
water forming a fine mist of sulfuric acid. This is a potential hazard and so absorption
into concentrated sulfuric acid is preferred.
Flow chart for the Contact Process
Sulfuric acid is also produced directly from sulfur dioxide - a by-product in metal production from sulfides.
When a metal sulfide such as zinc sulfide is roasted in air sulfur dioxide is formed.
2ZnS + 3O2 → 2ZnO + 2SO2
Concentrated sulfuric acid
Concentrated sulfuric acid is 98%, a heavy oily liquid, about twice as dense as water, and a dehydrating agent which reacts explosively with water unless great care is taken. When diluting concentrated sulfuric acid it must be added to water, slowly with stirring - and in the fume cupboard.
If you're doing as you oughta'
You add the acid to the water.
Sulfuric acid is the mother of all hygroscopic* liquids. Even dilute sulfuric reagent bottles must be tightly stoppered as sulfuric acid absorbs water vapour spontaneously from the atmosphere. Concentrated sulfuric acid will even extract water from compounds.
Blue copper sulfate crystals (hydrated copper sulfate, CuSO4.5H2O*), turn white when covered with concentrated sulfuric acid as white anhydrous copper sulfate (CuSO4*) is formed.
Carbohydrates like cellulose in cotton, wood and paper develop holes; starch and sucrose turn black.
(C6H10O5)n → 6nC + 5nH2O + heat
starch
C12H22O11 → 12C + 11H2O + heat
sucrose
A half a cup of white sugar crystals in a 100 mL beaker with concentrated sulfuric acid poured on top, rapidly turns volcanic in the fume cupboard with a porous black sticky column of carbon emerging from the beaker and setting as it cools into a rigid black mass.
Concentrated sulfuric acid is used as a drying agent. Chlorine gas produced by the electrolysis of NaCl is dried by passing it through concentrated sulfuric acid, as is the air used in the Contact Process.
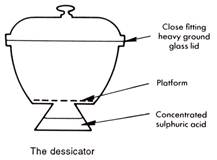
The apparatus used for drying substances and preventing hygroscopic* and deliquescent* materials from collecting moisture is the dessicator.
Poor misguided student We hear of him no more, For what he thought was H2O Was H2SO4.
*A glossary of terms is supplied at the end of the chapter
|
|
Dilute sulfuric acid
Dilute sulfuric acid is a strong acid; it reacts readily. Its chemistry can be divided into four main reactions.
(a) It will give an acid reaction to indicators.
(b) It will form carbon dioxide, CO2(g) with carbonates and bicarbonates.
Na2CO3(s) + H2SO4(aq) → Na2SO4(aq) + CO2(g) + H2O
2NaHCO3(s) + H2SO4(aq) → Na2SO4(aq) + 2CO2(g) + 2H2O
(c) It will form salts called sulfates with alkalis and metal oxides.
2NaOH(aq) + H2SO4(aq) → Na2SO4(aq) + 2H2O
CuO(s) + H2SO4(aq) + heat → CuSO4(aq) + H2O
(d) It will form hydrogen gas with active metals.
Zn(s) + H2SO4 → ZnSO4(aq) + H2(g)
Mg(s) + H2SO4(aq) → MgSO4(aq) + H2(g)
Sulfates
The salts of sulfuric acid are called sulfates. A salt is a chemical compound formed when a metal ion or an ammonium ion replaces a hydrogen atom in an acid.
Important sulfates include:
● Sodium sulfate, Na2SO4.10H2O, used in medicines
● Magnesium sulfate, MgSO4.7H2O, used in medicine as a laxative.
Epsom salts contain magnesium sulfate.
● Copper sulfate, CuSO4.5H2O, used in sprays to prevent fungus growth on vines and plants
● Aluminium sulfate Al2(SO4)3, used in the paper industry
● Barium sulfate, BaSO4 This is an insoluble sulfate and is given to patients in the form of a
suspension in water before X-rays are taken of the alimentary canal ( a 'barium meal').
● Calcium sulfate, CaSO4 This occurs in nature as gypsum, CaSO4.2H2O. It is slightly water soluble.
When gypsum is heated to 125oC it loses its water of crystallisation* and becomes plaster of Paris.
CaSO4.2H2O → CaSO4 + 3H2O
gypsum heat plaster of Paris
On adding water, the reaction is reversed and the crystals which form from the added water, interlock and form a hard mass. When plaster of Paris sets, it expands slightly. Because of this it is used to take impressions. For example dentists use it to get an exact impression of a patient's mouth. It is used to keep broken bones in place and it is used as a wall finish.
● Ammonium sulfate, (NH4)2SO4 used as a fertiliser (sulfate of ammonia).
The main use of sulfuric acid is in fertiliser production. It converts insoluble phosphate rock, Ca3(PO4)2 into mixtures that are water soluble. These are crushed and sold as 'super phosphate.'
Diprotic acids
Diprotic acids have two transferable protons and these can be transferred one at a time to form two kinds of salt.
With limited base added to sulfuric acid, hydrogen sulfates form. An example:
NaOH(aq) + H2SO4(aq) → NaHSO4(aq) + H2O
sodium hydrogensulfate
If the sodium hydroxide is present in sufficient quantity, sodium sulfate forms.
2NaOH(aq) + H2SO4(aq) → Na2SO4(aq) + 2H2O
Carbonic acid is another example of a diprotic acid. It forms hydrogencarbonates and carbonate salts.
Ammonia
Nitrogen is the second largest volume chemical produced globally. A major use is to make ammonia, NH3.
Ammonia is also a heavy chemical, and first produced synthetically in response to a looming global crisis of mass starvation in the early 20th century.
Plants need nitrogen to make plant protein, their DNA and RNA, and chlorophyll to trap sunlight so they can make food. Plants are the producers, the beginning of food chains, but elemental nitrogen N2 is inaccessible as a nutrient. Only lightning and very high temperatures have the energy to break the strong triple bond which makes elemental nitrogen so inactive. Soil bacteria can carry out this process too but by the end of the 19th century, there was not enough. Vast amounts of artificial fertiliser, containing 'fixed nitrogen' meaning water soluble nitrogen compounds accessible to plants were needed urgently. The challenge was to make it - to make ammonia - as ammonia and all ammonium salts are soluble in water.
Previous to this, from the 1840's to the 1870's guano dung mined off islands of Peru made it briefly the richest nation on Earth. The flow of income from this fertiliser provided three quarters of Peru's national budget until the wretched coolies who mined it hit bedrock. The next source was crystalline nitrates mined in South America's Atacama Desert. This could be purified into 'China Snow', the most important component of gunpowder. The nitrate wars started in 1879 but by 1881 Chile had sole control of the world's most valuable natural resource and Germany became its biggest customer by far. Germany's soil is poor, so Chile nitrate shipped from the other side of the world became its lifeblood.
Germany was a new nation and poor in resources, but rich in patriotism and scientific power. Enter Fritz Haber! He saw the atmosphere as a huge fertiliser silo and he set out to make fixed nitrogen by making ammonia. Unfortunately, temperatures high enough to atomise nitrogen destroyed any ammonia formed. A catalyst was needed. The catalyst was iron, but without aluminium oxide and calcium as 'promoters' it is ineffective. 200,000 materials were tested to achieve this result.
Haber achieved his success using higher pressures than those possible on a large scale. The giant chemical firm, BASF then employed the chemical engineer, Carl Bosch, who like Haber had the vision of feeding the world using fertiliser made from ammonia. Bosch not only achieved this with his innovative high pressure technology, but became head of BASF after World War I and saw its amalgamation into the huge industrial cartel, IG Farben. He and Haber had 'turbocharged' the process of converting elemental nitrogen into 'fixed nitrogen'
World War 1 was called 'The Chemists' War'. BASF at Oppau close to France was relocated in central Germany near Leuna and heavily defended. Ammonia production for fertiliser was switched to munitions production. Bosch's high pressure technology, exclusive to Germany was also used to produce methanol. Another IG Farben product was artificial rubber. Bosch modified the Bergius process to produce synthetic gasoline on a large scale by hydrogenation of coal. This was an enormous undertaking. Germany has no oil wells but vast deposits of coal. The hydrogen used to make ammonia was also feedstock for synthetic petrol. IG Farben was the largest business in Europe and the biggest chemical company in the world.
Haber who was Germany's foremost chemist and of Jewish descent, resigned in 1933 and Bosch who foretold the horrors to come, died in 1940. Both were Nobel laureates but their ideals had been betrayed. IG Farben became part of the Nazi machine in World War II making it self sufficient in rubber, explosives and fuel. In 1944, more than three quarters of the 35,000 workers were prisoners and slave labourers.
After the war, IG Farben was divided back into its component parts. BASF is the biggest Chemical company in the world but no longer a war machine and the fixed nitrogen output used again as Bosch originally intended. Haber-Bosch plants today consume 1-2 % of the world's energy supply and 3-5% of the world's natural gas production. It has been estimated that about half the nitrogen in our bodies today - the nitrogen in our protein, DNA and RNA, is synthetic and comes from Haber-Bosch factories. Without these it has been estimated that about 40% of the world's population would starve to death.
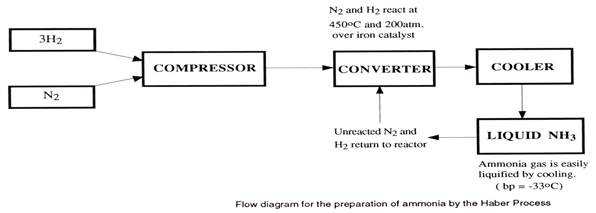
The Haber-Bosch Process Making ammonia in industry
The raw materials are: air, water and methane from natural gas.
Step 1 Hydrogen is produced by the reaction of methane with steam.
CH4(g) + H2O(g) → 3H2(g) + CO(g)
natural gas steam hydrogen carbon monoxide
Step 2 Nitrogen from air is produced by using some of this hydrogen to remove oxygen.
This is an exothermic reaction and the heat produced is used in step 1 where the temperature
required is about 1000oC.
2H2((g) + O2(g) → 2H2O(g)
Step 3 Carbon monoxide in step 1 is removed by first converting it to carbon dioxide using steam from
step 2. The steam produced is also used in step1.
H2O(g) + CO(g) → CO2(g) + H2(g)
Step 4 Only carbon dioxide is soluble in water and is dissolved under pressure.
Step 5 The nitrogen and hydrogen in a 1:3 ratio, are combined under pressure in the presence of a
catalyst to produce ammonia. This is the central step in the Haber-Bosch process.
N2((g) + 3H2(g) → 2NH3(g)
At a temperature of 450oC, 200 atmospheres pressure and with the special iron catalyst, about
15% conversion is achieved. Cooling the reaction mixture produces liquid ammonia and the
unreacted gases are recirculated.
Step 6 85% of ammonia produced today is used to make fertilisers. For example, the carbon dioxide
removed in step 3 is used to make urea from ammonia. Urea is a slow release fertiliser as it has
limited water solubility and so is favoured in tropical areas subjected to high rainfall. Urea is
another high bulk chemical.
2NH3 + CO2 → H2NCONH2 + H2O
urea
Flow chart for the Haber-Bosch Process
Making ammonia in the laboratory
Ammonia gas is made in the laboratory by two methods:
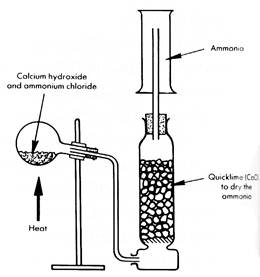
1 By heating a solid mixture of calcium hydroxide Ca(OH)2 and ammonium chloride NH4Cl.
Ca(OH)2(s) + 2NH4Cl(s) → CaCl2(s) + 2H2O(l) + 2NH3(g)
The gas is dried by passing it over calcium oxide which is deliquescent* and so an effective
drying agent.
2 By gently heating concentrated ammonia solution.
NH3(aq) → NH3(g)
The chemistry of ammonia
Ammonia is a base. It will react with acids to form salts. These salts are called ammonium salts.
When gaseous ammonia and gaseous hydrogen chloride meet, white fumes of ammonium chloride form.
NH3(g) + HCl(g) → NH4Cl(s)
Other ammonium salts are the fertilisers ammonium sulfate and ammonium nitrate.
● 2NH3(aq) + H2SO4 → (NH4)2SO4(aq) → (NH4)2SO4(s)
evaporate
● NH3(g) + HNO3(aq) → NH4NO3(aq) → NH4NO3(s)
evaporate
● All commercially produced nitric acid is made by the catalytic oxidation of ammonia.
4NH3 + 5O2 → 4NO2 + 6H2O
platinum-rhodium catalyst
2NO2 + H2O + ½O2 → 2HNO3
Synthetic fertilisers contain N, K and P, all in soluble form.
Properties of ammonia
1 Ammonia gas is colourless and has a pungent, choking odour. Small amounts stimulate respiration;
larger amounts cause paralysis of the respiratory system. Smelling the gas is to be avoided.
2 Ammonia gas is very soluble in water at 0oC and the most soluble gas known. One volume of water
will dissolve about 1100 volumes of ammonia. As the temperature increases, the solubility of ammonia
decreases. This is why solutions of ammonia always smell of ammonia gas. Solutions of ammonia are
sometimes labelled ammonia, short for ammonia solution. This solution is shown by the formula
NH3(aq).. Ammonia solution is alkaline, but it is not as strong an alkali as sodium hydroxide, NaOH.
3 Cooling and applying pressure liquefies ammonia gas easily.
4 Liquid ammonia is a good solvent. It will dissolve the alkali metals for example.
Tests for ammonia
1 Ammonia has a characteristic pungent smell.
2 Ammonia gas turns moist red litmus paper blue.
3 Ammonia gas forms solid white fumes with hydrogen chloride gas.
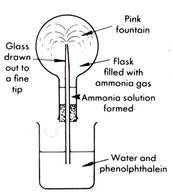
The fountain experiment
Ammonia's enormous solubility can be demonstrated with the fountain experiment.
Ammonia is made by the laboratory method 1 given on the previous page and as in the diagram below.
Assemble the apparatus as shown in the second diagram but do not put together. The flask must be dry. Alternatively use concentrated ammonia solution by itself. (This is easier).
Method 1 Gently heat the reagents - the drying tower can be omitted on a dry day. 2 The gas must be collected in the dry round-bottomed flask. 3 A moist piece of red litmus paper placed at the mouth of the flask will turn blue when the jar is full.
|
|
|
Method (work quickly!) 1 With the filled container of ammonia, insert the glass tube and stopper and place as shown in the diagram. 2 Gently heat the flask for 3 seconds by moving a burner over its surface. This will cause the gas to expand. Remove the burner. The gas will contract and the water will rise in the tube.
What does the change in colour of the indicator tell you about the nature of the ammonia solution?
|
Glossary
Hygroscopic
Hygroscopic solids extract water vapour spontaneously from the atmosphere.
e.g. Sweet biscuits become soft, boiled sweets become sticky and paper swells, jamming photocopying machines. Hair too is hygroscopic.
Hygroscopic liquids spontaneously absorb water vapour from the atmosphere, diluting themselves.
e.g. methanol, ethanol and glycerol.
Sulfuric acid extracts H & O in the ratio 2:1 from carbohydrates like cane sugar so that C12H22O11 is converted exothermically to black carbon and holes form in paper and cotton lab coats. (They're made from cellulose, also a carbohydrate).
Deliquescent
This is a term reserved for solids which spontaneously absorb water vapour from the atmosphere, eventually forming a solution.
e.g. magnesium sulfate, MgSO4, calcium chloride, CaCl2 and sodium hydroxide, NaOH, are all deliquescent. Sodium hydroxide, being a base, also dissolves atmospheric carbon dioxide, reacting to form sodium hydrogencarbonate (also called sodium bicarbonate, NaHCO3) and sodium carbonate, Na2CO3..
Water of Crystallisation
The correct formula for blue copper sulfate is CuSO4.5H2O. Without the chemically combined water, copper sulfate would not be blue or crystalline. This chemically combined water is called water of crystallisation.
Blue copper sulfate is called hydrated copper sulfate or copper sulfate pentahydrate or even copper sulfate five water!
Do all salts have water of crystallisation? No, common salt, NaCl, does not.
Heating hydrated salts decomposes them. Chemical bonds are broken, and so this is an endothermic chemical change.
e.g. CuSO4.5H2O(s) → CuSO4(s) + 5H2O(l) The colour change is from blue to white.
and CoCl2.6H2O(s) → CoCl2(s) + 6H2O(l) The colour change is from pink to blue - very pretty
CuSO4 is called white copper sulfate or anhydrous copper sulfate. It is hygroscopic.
A test for the presence of water in a mixture is to drop some on white anhydrous copper sulfate. A positive test is if the white solid turns blue.
Efflorescence
Solids which are efflorescent or show efflorescence are those which spontaneously lose water of
crystallisation to the atmosphere and so become powdery.
e.g Na2CO3.10H2O → Na2CO3. (10 - x).H2O The composition becomes uncertain.
Exercises
1 Copy and complete the following table.
Name of molecule |
Molecular formula |
Electron dot structure |
Valence bond structure |
Name of shape |
Polar or non-polar? |
nitrogen |
|
|
|
|
|
water |
|
|
|
|
|
ammonia |
|
|
|
|
|
Sulfur trioxide |
|
|
|
|
|
Sulfuric acid |
|
|
|
|
|
2 Describe a test:
(a) to show the presence of water in a mixture
(b) to show that a sample of water is pure.
3 Gases can be dried, (that is, the impurity water vapour is removed) by bubbling them through concentrated
sulfuric acid. Oxygen can be purified this way but not ammonia. Can you explain why?
4(a) Write down the key equation for the industrial preparation of ammonia.
(b) How could you:
(i) demonstrate its extreme solubility in water?
(ii) demonstrate that it is basic?
(c) Write down an equation to show how you could prepare ammonium sulfate, starting from ammonia.
Source : http://www.usc.adelaide.edu.au/local/transitionlectures/chemistry/industrialchemistry.doc
Web site: http://www.usc.adelaide.edu.au
Google key word : Industrial chemistry and some important covalent file type : doc
Author : not indicated on the source document of the above text
If you are the author of the text above and you not agree to share your knowledge for teaching, research, scholarship (for fair use as indicated in the United States copyrigh low) please send us an e-mail and we will remove your text quickly.
Industrial chemistry and some important covalent
If you want to quickly find the pages about a particular topic as Industrial chemistry and some important covalent use the following search engine:
Chemistry
Industrial chemistry and some important covalent
Please visit our home page
Larapedia.com Terms of service and privacy page